Our aim in this research note is to compare curcumin and clobetasol propionate in management of patients with oral lichen planus. Sixty- four patients of OLP were divided where 32 patients were put in group I in which patients received 0.1% triamcinolone and in group 2, 32 patients received 5% curcumin paste for 4 weeks. Measurement of the appearance score and severity of pain was done at baseline and at the end of 2 and 4 weeks. We found that complete remission in pain was seen in 30% in group 1 and 35% in group 2, good response in 28% in group 1 and 17% in group 2, poor response in 24% in group 1 and 25% in group 2 and no response in 18% in group 1 and 33% in group 2. A non- significant difference among good and no response was seen (P<0.05). Oral lichen planus is common inflammatory autoimmune disease. Curcumin in our study showed slightly better results than triamcinolone, hence suggesting that it can be used in patients with oral lichen planus.
Oral lichen planus (OLP) is a chronic mucocutaneous, autoimmune and inflammatory disease affecting oral mucosa, skin and genitalia which is T-cell mediated [1,2]. It involves 0.1-2% of general population. It has malignant potential between 0.07% and 5.8%. It commonly affects females in their 4th decade of life and clinically appears as white striations, erosions and desquamative gingivae [3]. Various forms are reticular, erosive, erythematous, popular, plaque, bullous, annular and ulcerative [4]. Main pathogenesis of OLP is antigen specific cell mediated immune response, which includes accumulation of activated CD8+lymphocytes on basal keratinocytes causing apoptosis. Genetic, psychological factors initiate autoimmune mediated response [5].
Corticosteroids are the mainstay treatment modality and effective in controlling the symptoms of the disease; but side effects such as secondary candidiasis, telangiectasia, hypothalamic-pituitary-adrenal suppression, muco-cutaneous atrophy and increased potential of systemic absorption are also common with its prolonged use [6]. Curcuma longa is a perennial plant belonging to Zingiberaceae family possessing anti-inflammatory effects. Its main constituents include three curcuminoids including curcumin (the primary ingredient and the one responsible for its yellow color and anti-inflammatory effect), demethoxycurcumin and bisdemethoxycurcumin [7]. It has anti-oxidant, anti-carcinogenic, anti-microbial, anti-proliferative and wound healing properties. Studies show that curcumin reduces multiple sclerosis, rheumatoid arthritis, psoriasis, inflammatory bowel disease in human and animal models [8]. These properties have prompted investigators to check its efficacy on oral diseases namely, lichen planus, oral submucous fibrosis (OSMF), leukoplakia, recurrent aphthous stomatitis, etc. Previous studies have indicated that use of topical curcumin has a chemopreventive effect in oral potentially malignant disorders such as OSMF and leukoplakia [9,10]. Considering this, we attempted this study to compare curcumin and clobetasol propionate in management of patients with oral lichen planus.
A simple stratified random sampling was performed where 32 patients were put in group I in which patients received 0.1% triamcinolone and in group 2, 32 patients received 5% curcumin paste for 4 weeks and were asked to apply it TDS per day after eating and brushing. All patients were refrain from eating for 20 minutes. Measurement of the appearance score and severity of pain was done at baseline and at the end of 2 and 4 weeks and recorded in the patients’ questionnaires. In our study we used the criteria by Thongprasom et al. for determining the appearance score as: 0: No lesion, normal mucosa, 1: Mild white striae, no erythematous area, 2: White striae with atrophic area less than \(1cm^{2}\),3: White striae with atrophic area more than \(1cm^{2}\) , 4: White striae with ulcerative area less than \(1cm^{2}\) and 5: White striae with ulcerative area more than \(1cm^{2}\). Response rate was measured as complete remission-100% reduction in signs and symptoms, good response- 50% or more reduction in signs and symptoms and poor response- < 50% reduction in signs and symptoms. Results were compared in two groups using Mann Whitney U test where p value less than 0.05 considered significant.
The mean age was 45.2 years in group 1 and 43.7 years in group 2. Males: Females ratio of 10:22 and 11:21 was seen in group 1 and 2 respectively. Pain at baseline (VAS) was 5.48 in group 1 and 5.12 in group 2. At baseline, lesion size was 3.85 cm2 in group 1 and 3.72 \(cm^{2}\) in group 2, see Table 1.
| Characteristics | Group 1 | Group 2 |
|---|---|---|
| Mean age (years) | 45.2 | 43.7 |
| M:F | 10:22 | 11:21 |
| Pain at baseline (VAS) | 5.48 | 5.12 |
| Lesion size at baseline (cm2) | 3.85 | 3.72 |
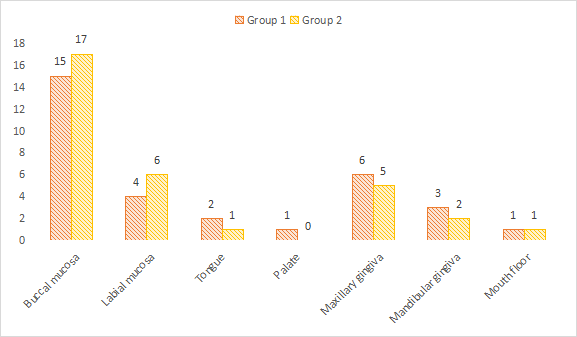
Common site was buccal mucosa in both groups (group 1- 15, group 2- 17), maxillary gingiva (group 1- 6, group 2- 5), labial mucosa (group 1- 4, group 2- 6), mandibular gingiva (group 1- 3, group 2- 2), tongue (group 1- 2, group 2- 1), floor of mouth (group 1- 1, group 2- 1) and palate (group 1- 1, group 2- 0). A non- significant difference among site distribution was seen (P> 0.05), see Table 2 and Figure 1.
| Site | Group 1 | Group 2 | P value |
|---|---|---|---|
| Buccal mucosa | 15 | 17 | >0.05 |
| Labial mucosa | 4 | 6 | |
| Tongue | 2 | 1 | |
| Palate | 1 | 0 | |
| Maxillary gingiva | 6 | 5 | |
| Mandibular gingiva | 3 | 2 | |
| Mouth floor | 1 | 1 |
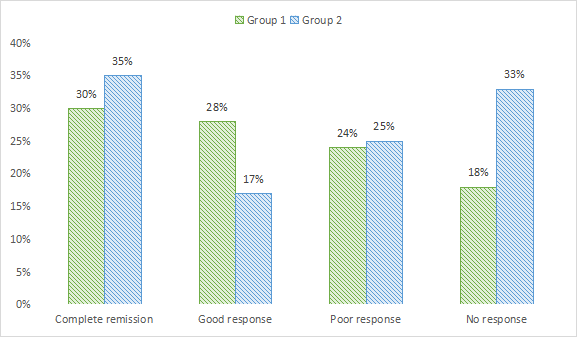
Complete remission in pain was seen in 30% in group 1 and 35% in group 2, good response in 28% in group 1 and 17% in group 2, poor response in 24% in group 1 and 25% in group 2 and no response in 18% in group 1 and 33% in group 2. A non- significant difference among good and no response was seen (P< .05), see Table 3 and 2.
| Response | Group 1 | Group 2 | P value |
|---|---|---|---|
| Complete remission | 30% | 35% | >0.05 |
| Good response | 28% | 17% | < 0.05 |
| Poor response | 24% | 25% | >0.05 |
| No response | 18% | 33% | < 0.05 |
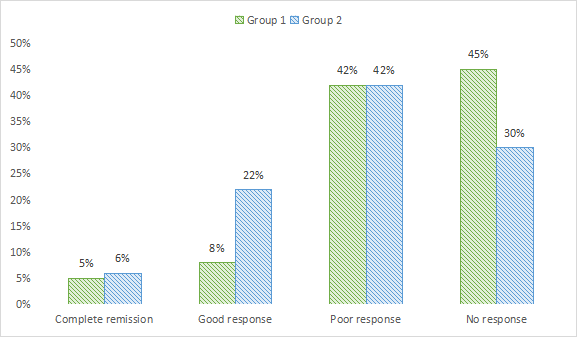
Complete remission in clinical response was seen in 5% in group 1 and 6% in group 2, good response in 8% in group 1 and 22% in group 2, poor response in 42% in group 1 and in group 2 and no response in 45% in group 1 and 30% in group 2. A non- significant difference among good and no response was seen (P< .05), see Table 4 and Figure 3.
| Response | Group 1 | Group 2 | P value |
|---|---|---|---|
| Complete remission | 5% | 6% | >0.05 |
| Good response | 8% | 22% | < 0.05 |
| Poor response | 42% | 42% | >0.05 |
| No response | 45% | 30% | < 0.05 |
Our study showed that the mean age was 45.2 years in group 1 and 43.7 years in group 2. There were10 males and 22 females in group 1 and 11 males and 21 females in group 2. Dharman et al., [14] study comprised of 315 OLP patients, wherein four studies revealed that topical curcumin had no statistically significant difference when compared to corticosteroids in treating pain, burning sensation, erythema and ulceration. In three clinical trials compared with placebo, one study showed statistical significance with increased oral dosage (6000 mg) of curcumin, two were not significant due to its reduced oral dosage (2000 mg). Two studies showed that curcumin was effective with increased concentration. Three studies with no controls were statistically significant in reducing burning sensation and clinical appearance of OLP.
Our study demonstrated that pain at baseline (VAS) was 5.48in group 1 and 5.12 in group 2. At baseline, lesion size was 3.85 cm2 in group 1 and 3.72 \(cm^{2}\) in group 2. Keshari et al., [16] assessed the efficacy and safety of topical curcumin in the management of OLP. 27 adult OLP patients, who were randomly divided into two groups. The control group (n = 12) was treated with triamcinolone acetonide 0.1% and the study group (n = 15) with commercially available topical curcumin ointment each to be applied thrice daily for 2 weeks. The patients were reviewed every week. The research groups showed a significant reduction in all the parameters measured. The comparison showed significant improvement in the erythema (P = 0.002), but non-significant reduction in pain (P = 0.697), and ulceration (P = 0.291) in the study group as compared to the control group.
Our findings were common site was buccal mucosa in group 1- 15 and group 2- 17, maxillary gingiva in group 1- 6 and group 2- 5, labial mucosa in group 1- 4, group 2- 6, mandibular gingiva in group 1- 3, group 2- 2, tongue in group 1- 2, group 2- 1, floor of mouth in group 1- 1, group 2- 1 and palate in group 1- 1, group 2- 0.
Thomas AE et al., [17] assessed increased reduction of burning sensation measured by NRS, observed in triamcinolone acetonide group, followed by curcumin 6 times daily application. Curcumin oral gel, applied 3 times a day, was insignificant compared to TA. Application of curcumin oral gel six times daily was considered equally effective compared to triamcinolone acetonide. There was comparable effect in pain reduction score in both triamcinolone and curcumin groups with non- statistical significant difference between two groups. Effects were similar to those of topical corticosteroids, and thus, it can be a suitable alternative to corticosteroids.
Chainani-Wu et al., [18] administered a primary dose of systemic prednisone (60 mg/day for the first week) in both groups, which was not done in our study and could have affected their final results. Long periods of observation not only can interfere with patient compliance and cooperation. Curcumin, a phytochemical has been used as a dietary supplement as well as a therapeutic agent. Curcumin displays a wide range of pharmacological properties against various human disorders. Curcumin exhibits strong anti-inflammatory, analgesic, antioxidant, anti-aging, chemopreventive, antitumoral, anti-angiogenic, anti-metastatic, radio sensitizing, and chemosensitizing effects in cancer. Of therapeutic interest, studies have indicated that curcumin as a lone therapeutic agent is safe and exhibits no major toxicity [19].